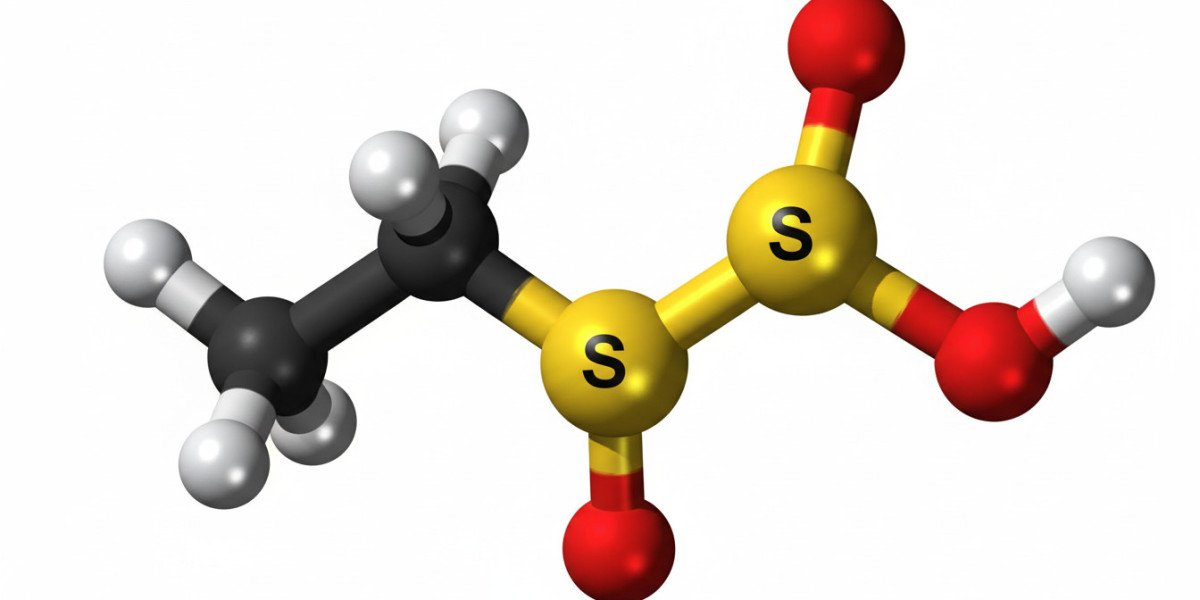
Ethylene sulfate is a versatile chemical compound widely used in various industrial applications. It is an organic sulfate ester derived from ethylene glycol. Due to its reactive nature, ethylene sulfate serves as an important intermediate in chemical synthesis, especially in the production of surfactants, detergents, and pharmaceuticals. Understanding its properties, applications, and safety considerations is essential for industries that utilize this compound.
Chemical Structure and Properties
The chemical formula of ethylene sulfate is C2H4SO4. It exists as a colorless or slightly yellow crystalline solid under standard conditions. Its molecular structure features a cyclic sulfate group, which makes it highly reactive with nucleophiles such as water and alcohols. Ethylene sulfate has a relatively low melting point and is soluble in water and polar organic solvents. Its reactivity is attributed to the strained three-membered ring in its structure, which can undergo ring-opening reactions in various chemical processes.
Industrial Production
Ethylene sulfate is primarily produced by reacting ethylene oxide with sulfuric acid. This reaction yields a cyclic sulfate compound, which can then be purified for industrial use. The production process requires careful control of temperature and reaction time to prevent decomposition and side reactions. Industrial-scale production facilities implement rigorous safety protocols due to the highly reactive and potentially hazardous nature of the intermediates.
Applications in Chemical Industry
Surfactants and Detergents
One of the main applications of ethylene sulfate is in the production of surfactants and detergents. It is a key intermediate in the synthesis of ethoxylated sulfates, which are widely used in household and industrial cleaning products. These compounds provide excellent foaming properties and help remove grease and dirt efficiently.
Pharmaceutical Applications
Ethylene sulfate also plays a role in pharmaceutical synthesis. It is used to introduce sulfate groups into drug molecules, improving their solubility and bioavailability. Additionally, some specialty chemicals derived from ethylene sulfate are utilized in the development of active pharmaceutical ingredients (APIs).
Polymer and Resin Production
In the polymer industry, ethylene sulfate is used as a precursor for water-soluble polymers and resins. These polymers have applications in adhesives, coatings, and paper treatment. The sulfate functionality improves polymer solubility and ionic properties, making them suitable for diverse industrial formulations.
Environmental and Safety Considerations
Handling ethylene sulfate requires strict adherence to safety guidelines. The compound is corrosive and can cause severe irritation to the skin, eyes, and respiratory system. Industrial facilities employ personal protective equipment (PPE) and specialized storage conditions to minimize risk. Accidental release of ethylene sulfate into the environment can be harmful, as it reacts with water to form acidic solutions that may affect soil and water systems. Proper disposal and containment strategies are therefore critical.
Recent Research and Innovations
Recent studies have focused on developing safer and more efficient methods for synthesizing ethylene sulfate derivatives. Green chemistry approaches aim to reduce the use of hazardous reagents and minimize waste generation. Additionally, researchers are exploring the use of ethylene sulfate in advanced materials, including biodegradable polymers and specialty surfactants with improved environmental profiles.
Conclusion
Ethylene sulfate is a highly reactive chemical compound with significant industrial importance. Its applications range from detergents and pharmaceuticals to polymers and specialty chemicals. While it offers numerous benefits, handling it safely is paramount due to its corrosive nature and environmental impact. Ongoing research continues to expand its potential uses while prioritizing safer and more sustainable production methods.